Na-ion batteries are promising in large-scale energy storage owing to the abundant raw material resources, low cost, and high safety.
Sodium vanadium fluorophosphate (Na3(VOPO4)2F) with a theoretical energy density of 480 Wh/Kg is regarded as a strong candidate among various cathode materials. However, the intrinsic low conductivity and high energy-consumption during synthesis process hinder its commercialization.
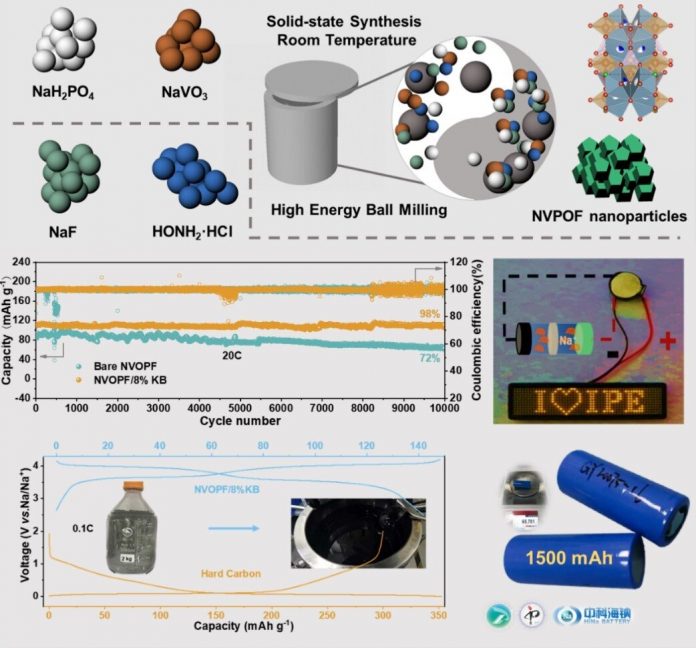
Researchers from the Institute of Process Engineering (IPE) and the Institute of Physics of the Chinese Academy of Sciences developed a one-step mechanochemical method to rapidly prepare the polyanionic compound sodium vanadium fluorophosphate as the cathode materials for Na-ion batteries, which exhibited excellent rate performance and cycle stability.
The prepared Na3(VOPO4)2F/KB composite delivered a high discharge capacity of 142 mAh g-1 at 0.1C. The extra capacity beyond the theoretical specific capacity (130 mAh g-1) benefited from the interfacial charge storage.
Moreover, a specific capacity of 112 mAh g-1 can be obtained even at 20 C, which means this Na-ion battery could be fully charged/discharged in three minutes.
Superior cycle stability of this composite was demonstrated by ultrahigh cycling stability with 98% retention over 10,000 cycles.
High-resolution transmission electron microscopy revealed that the nanocrystallines of Na3(VOPO4)2F about 30 nm were embedded in the carbon framework, which facilitated the rapid conduction of electrons and Na ions.
The reversible structural evolution and negligible volume change of Na3(VOPO4)2F/KB composite during charging/discharging were also demonstrated by in situ X-ray diffraction and 23Na nuclear magnetic resonance spectrum.
“The method provides a feasible strategy to improve the rate performance and cycle performance of cathode materials. Besides, the kilogram-scale product indicates the mechanochemical method is suitable for rapid large-scale production electrode materials for Na-ion batteries,” said Prof. Zhao Junmei, a co-corresponding author of the study.